MATERIALS SCIENCE AND CHEMISTRY
Crystallization of Amorphous Phase Change Memory Material: Molecular Dynamics Simulations
Principal Investigator:
Robert O. Jones
Affiliation:
Forschungszentrum Jülich (Germany)
Local Project ID:
hfz01
HPC Platform used:
JUQUEEN of JSC
Date published:
The phase change materials (PCM) used in optical storage devices (DVD, Blu-ray Disc, etc.) must show a very rapid transition between the different phases of the recording layers of optical memories. The data “bits” are tiny amorphous marks formed by laser melting and subsequent quenching in a thin amorphous layer, and the amorphous marks can be recrystallized (“erased”) by laser annealing below the melting point. To be of practical use, the time scale should be around 10 nanoseconds or less, which is satisfied by very few materials indeed. Commercial storage materials are almost always alloys of antimony and tellurium, often with additional elements, such as germanium, indium, or silver. The essential challenge facing the phase-change industry for over 20 years can be stated simply: How can we reduce the crystallization time of the amorphous bits? This cannot be answered unless the structures of the amorphous and crystalline states are known and the mechanism of the transition between them is clarified.
These questions can be addressed by molecular dynamics simulations on the basis of density functional (DF) theory. DF calculations are free of adjustable parameters and have given useful results and valuable predictions in many contexts. Nevertheless, they are numerically very demanding and must be carried out here for a large sample (hundreds of atoms) and repeated very many times in order to simulate a process as complex as crystallization. In the case of phase change materials, the process is very rapid (some nanoseconds), but about one million times longer than the time step in the simulations, so it is necessary to perform millions of complicated calculations to achieve the goal.
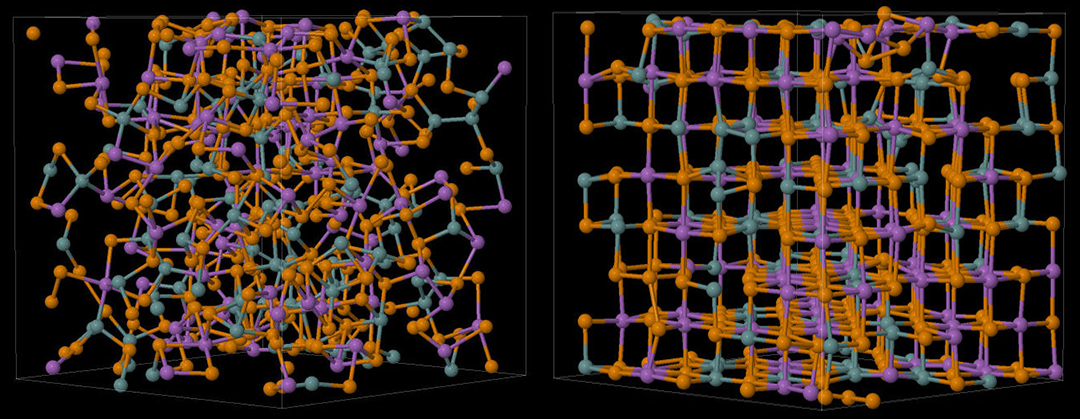
Supercomputers are absolutely essential for this task, and the work at the Forschungszentrum Jülich has used the GCS supercomputer JUQUEEN. The goals of this research have been to understand the structure of the amorphous state and to determine the processes essential to its crystallization. The first goal was achieved some time ago [1], where it as demonstrated that the amorphous and crystalline structures of Ge2Sb2Te5 (GST), a prototype PCM used in commercial memory devices, shared a particular structural motif (see left frame of Fig. 1), namely four-membered rings alternating between Te atoms and Ge or Sb atoms.
The most recent results provide insight into the dynamics of crystallization of GST. Four simulations of the same initial structure at 600 K showed varying degrees of crystallization [2,3]. The sample with the most complete crystallization is shown in Fig. 1 (right frame), and it was exciting to follow the process that took the disordered structure [Fig. 1, left frame] to the ordered structure, which has a rock salt form, with Te atoms on one sublattice and a random mixture of Ge and Sb atoms and vacancies on the other.
In contrast to experimental scattering studies (neutrons, x-rays), such simulations provide direct information about the structure and the way it changes. This information should provide essential insight into ways to design alloys with even better properties (more rapid crystallization, lower crystallization temperature and power requirements) and lower production costs.
Publications:
[1] J. Akola; R. O. Jones : Structural phase transitions on the nanoscale: The crucial pattern in the phase-change materials Ge2Sb2Te5 and GeTe
[Online-Edition: http://dx.doi.org/10.1103/PhysRevB.76.235201]
In: Phys. Rev. B 76, 235201 (2007)
[2] J. Kalikka; J. Akola; R. O. Jones : Simulation of Crystallization in Ge2Sb2Te5: A memory effect in the canonical phase-change material
[Online-Edition: http://dx.doi.org/10.1103/PhysRevB.90.184109]
In: Physical Review B, 90, 184109 (2014)
[3] J. Kalikka; J. Akola; R. O. Jones : Crystallization processes in the phase change material Ge2Sb2Te5: Unbiased density functional/ molecular dynamics simulations
[Online-Edition: http://dx.doi.org/10.1103/PhysRevB.94.134105]
In: Phys. Rev. B 94, 134105 (2016)
Research Team:
Jaakko Akola, Janne Kalikka (Tampere University of Technology, Finland)
Scientific contact:
Robert O. Jones
Forschungszentrum Jülich, Peter Grünberg Institut PGI-1
D-52425 Jülich, Germany
e-mail: r.jones [@] fz-juelich.de
November 2016